
US Northwestern University New cancer treatment: crossing the blood-brain barrier
The Proceedings of the National Academy of Sciences has published online a study entitled "New treatment extends breast cancer survival, prevents recurrence, extends life of treated mice with triple-negative breast cancer by 150%". Researchers at Northwestern University (USA) have developed a new immunotherapy treatment that significantly extends the survival time of mice with triple-negative breast cancer. Triple-negative breast cancer is one of the most aggressive and difficult to treat types of breast cancer. In a new study, mice that received this therapy had complete remission of their tumours for at least 100 days. All untreated mice died at day 30. None of the treated mice experienced adverse side effects or autoimmune reactions. Chad A. Mirkin, the George B. Rathmann Professor of Chemistry in the Weinberg College of Arts and Sciences at Northwestern University, Director of the International Institute for Nanotechnology and a member of the Robert H. Lurie Comprehensive Cancer Centre at Northwestern University, led this research. Chad, who led the research and invented SNA, credits the shape and structure of the nanoparticles with the success of immunotherapy.
Nanotherapies are spherical nucleic acid (SNA) drugs with a unique 3D design, consisting of nanoparticles surrounded by nucleic acid, say industry sources. The drug crosses the blood-brain barrier and causes apoptosis of tumour cells. Glioblastoma is a fatal brain tumour that affects thousands of patients in the United States each year; there is no effective treatment for this deadly brain cancer. Initial clinical trials in patients with recurrent glioblastoma have shown that small doses are not harmful to humans and that the drug does cross the blood-brain barrier. A new class of drugs using the SNA platform could be applied to other types of neurological disorders, including Alzheimer's and Parkinson's, by crossing the blood-brain barrier and targeting the tumour cells associated with these diseases.
Cannabinoids may play a big role in effectively crossing the blood-brain barrier and causing apoptosis of tumour cells.
The news instantly got Northwestern University very much in the spotlight, with people frantically searching for solutions to fight cancer, and is there a better way to break the blood-brain barrier or not?
Yes, let's not forget that breaking the blood-brain barrier, cannabinoids may play a big role! The body has built a blood-brain barrier to protect the brain, preventing harmful substances from entering the brain tissue from the bloodstream and protecting the central nervous system, but the problem that this brings is that many drugs cannot enter the brain once it is sick. Since cannabinoids can pass the blood-brain barrier, we can use it as a medium to develop new drugs to treat brain diseases by loading small molecules and other drugs on it. So, the FDA's approval of the world's first CBD drug for the treatment of childhood epilepsy on June 26, 2018, on International Drug Day, only opened the tip of the iceberg for the medical use of industrial hemp.
GW Pharmaceuticals (GWPH), a leading company in the cannabis industry, is already on its way to winning approval for the first ever cannabis-derived therapy in the United States. But an early trial suggests that these treatments may also be an effective way to fight one of the most devastating forms of brain cancer: glioblastoma multiforme. The UK-based company published preliminary data from a mid-stage study back in 2017 on an experimental drug combining cannabidiol and THC. The results so far showed that the drug improved the median survival rate of brain cancer patients by about six months compared to a placebo. Typically, this type of cancer destroys the brain and kills (on average) 70% of patients within two years of being diagnosed.
"We believe that the efficacy signals shown in these studies further strengthen the potential role of cannabinoids in oncology and offer GW the prospect of a new and unique cannabinoid product candidate for the treatment of glioma," Justin Gover, GW CEO, said in a statement.
How do cannabinoids cross the blood-brain barrier and cause apoptosis in tumour cells?
In fact, the particular efficacy of cannabinoids in the treatment of cancer is something that should be put in the spotlight, studied by the world and remembered by the world. A very detailed review of cannabinoid treatment of cancer has been published in the American Journal of Immunobiology back in August 2010 in the article "Cannabinoid-induced apoptosis in inmune cell as a pathway to immunosuppression."
Cannabinoids are a group of compounds found in the cannabis plant (Cannabis sativa L.) that regulate their physiological and behavioural effects by activating specific cannabinoid receptors. With the recent discovery of cannabinoid receptors (CB1 and CB2) and endogenous cannabinoid systems, the field of cannabinoid research has expanded exponentially. Cannabinoids have been shown to act as potent immunosuppressive and anti-inflammatory agents and have been shown to mediate beneficial effects in a variety of immune-mediated diseases such as multiple sclerosis, diabetes, infectious shock, rheumatoid arthritis and allergic asthma. Cannabinoid receptor 1 (CB1) is mainly expressed in cells of the central nervous system as well as in peripheral cells. In contrast, cannabinoid receptor 2 (CB2) is mainly expressed on immune cells. The precise mechanisms by which cannabinoids mediate immunosuppression are only just beginning to be understood and can be broadly classified into four pathways: apoptosis, inhibition of proliferation, inhibition of cytokine and chemokine production, and induction of T regs (T regulatory cells). This article focuses on the mechanisms by which natural and synthetic cannabinoids induce apoptosis through activation of CB2 receptors, focusing on the mechanisms of cannabinoid-mediated immunosuppression of apoptosis in different immune cell populations, and discusses how activation of CB2 offers a new therapeutic modality for inflammatory and autoimmune diseases and immune system malignancies without adverse psychotropic effects.
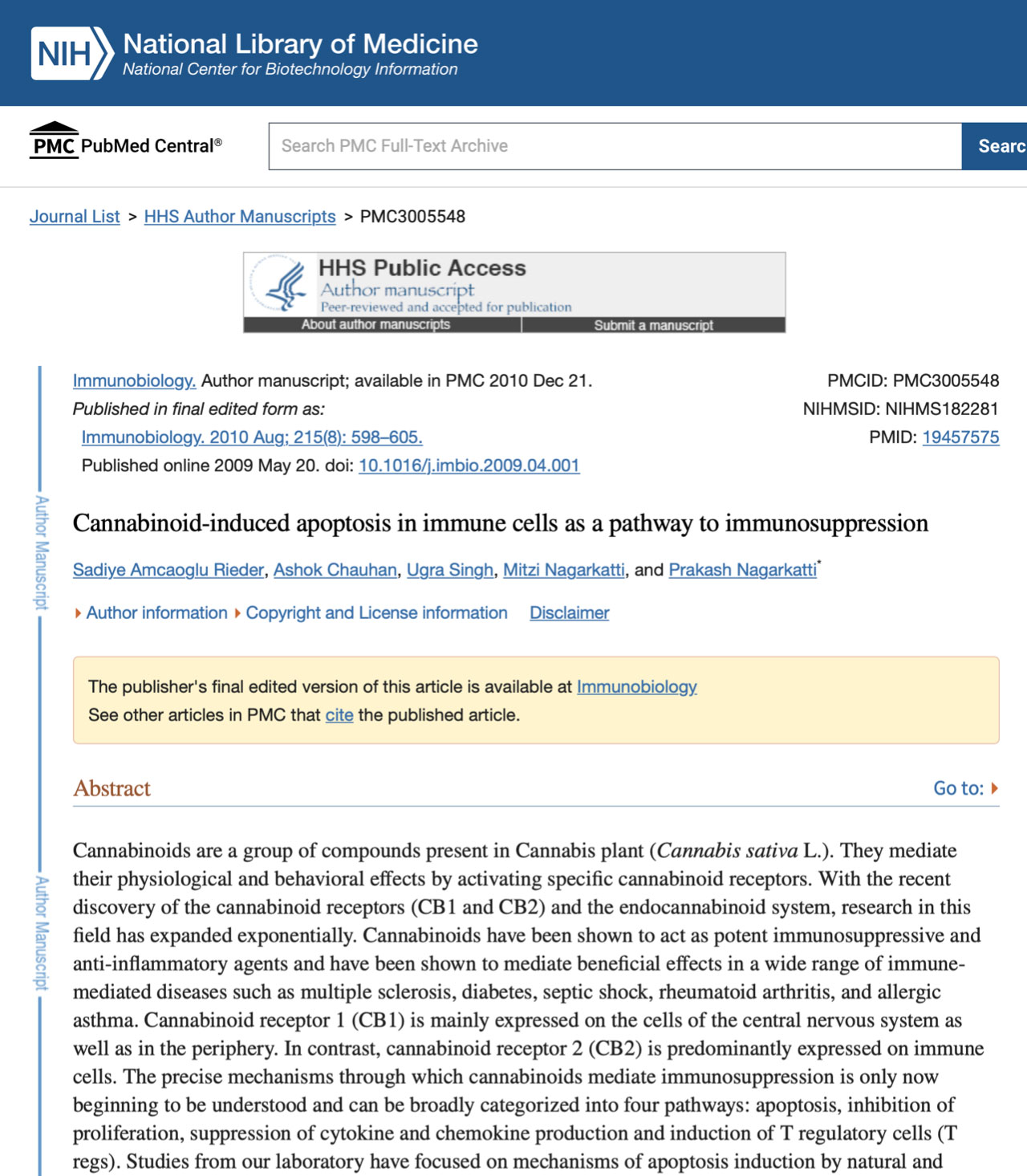
Cannabis has been used for centuries for recreational and medicinal purposes. Cannabinoids have been shown to be effective in the treatment of nausea and vomiting associated with cancer chemotherapy, anorexia and cachexia in HIV/AIDS patients, and neuropathic pain and spasticity in multiple sclerosis (Guzman 2003; Tramer et al. 2001; Inui 2002; Pollmann and Fenenberg 2008). Recently, the anti-inflammatory properties of cannabinoids have attracted considerable attention. Over the past 15 years, research on cannabinoids has led to the discovery of cannabinoid receptors (CB1 and CB2) and their endogenous ligands, which constitute the endocannabinoid system.CB1 and CB2 are both heterotrimeric Gi/o protein-coupled receptors.CB1 is expressed in both central nervous system (CNS) and peripheral tissues, mainly on presynaptic nerves.CB2, sometimes referred to as the "peripheral cannabinoid", is a heterotrimeric receptor. CB2, sometimes referred to as the 'peripheral cannabinoid receptor', is mainly expressed on immune cells, but can also be found in other peripheral tissues such as the retina and CNS (Croxford and Yamamura 2005; Mackie 2006). The endocannabinoid system regulates many functions, such as movement, memory, appetite, thermoregulation, pain and immune function. Natural and synthetic cannabinoids are derived from the cannabis plant, while endocannabinoids are metabolites of arachidonic acid (Klein and Cabral 2006). The picture summarises some of the well-known natural and synthetic cannabinoids, as well as their sources and receptors.
In addition, a group of researchers studied the effects of the non-psychoactive cannabinoid cannabidiol (CBD) on apoptosis in immune cells (Lee et al., 2008). They demonstrated that CBD induced apoptosis in a time- and dose-dependent manner in CD4+ and CD8+ T cell populations. Treatment of splenocytes with 4-8 μM CBD significantly increased apoptosis, as determined by the percentage of subdiploid and TUNEL-positive cells. In addition, the researchers showed that apoptosis was due to the formation of reactive oxygen species (ROS) and the activation of cystein 8 and cystein 3. The same group also reported that CBD had similar effects on mouse thymocytes and EL-4 cells.
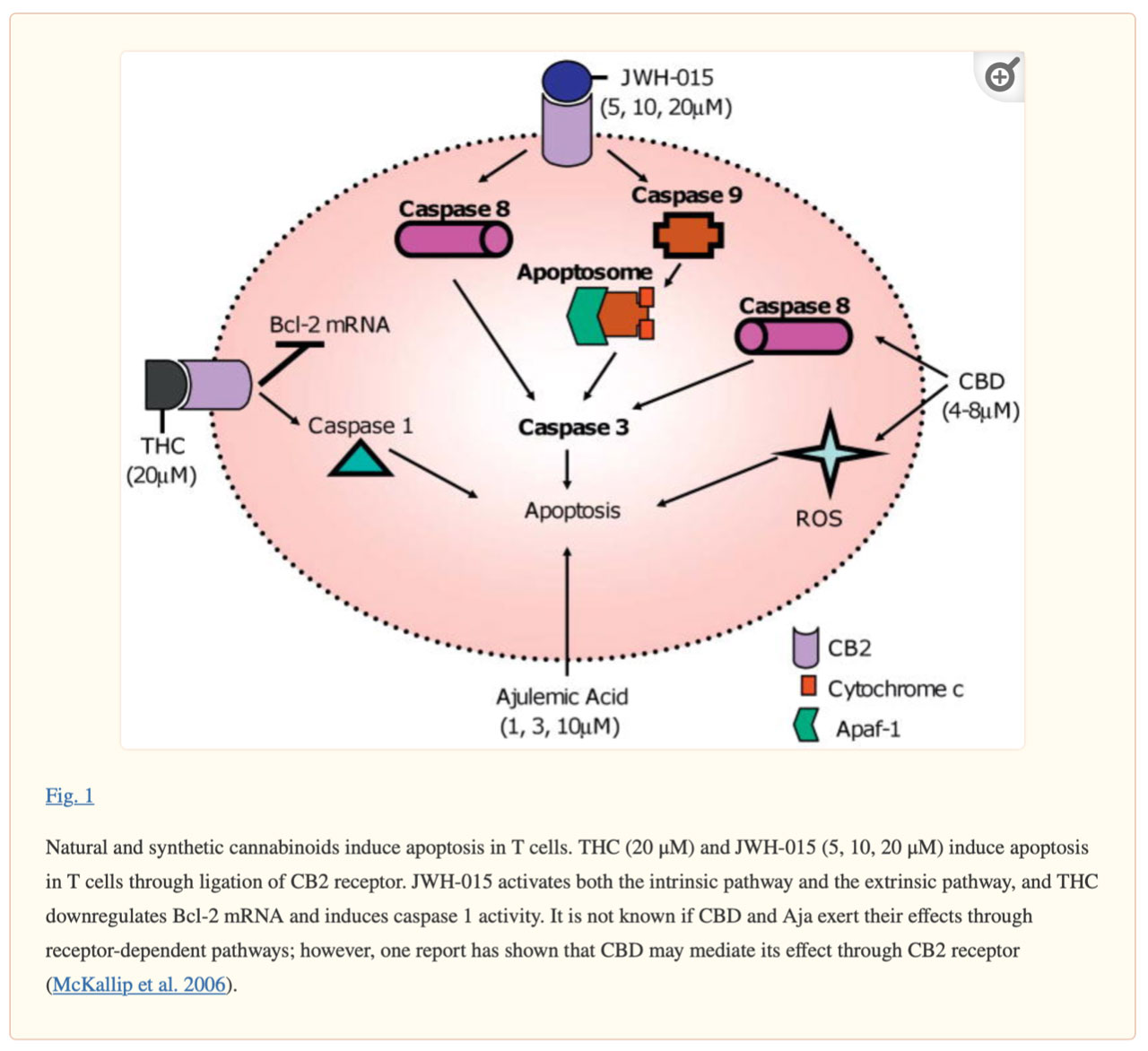
Natural and synthetic cannabinoids induce apoptosis in T cells. THC (20 μM) and JWH-015 (5, 10, 20 μM) induce T cell apoptosis by linking CB2 receptors. jWH-015 activates both intrinsic and extrinsic pathways, and THC downregulates Bcl-2 mRNA and induces caspase 1 activity. It is unclear whether CBD and Aja act through a receptor-dependent pathway; however, one report suggests that CBD may mediate its action through the CB2 receptor (McKallip et al., 2006).
Cannabinoids have also been used globally, including in the treatment of some cancers, and since they can effectively penetrate the blood-brain barrier, they must be at the top of the list for the treatment of the brain. Of course there is a wider range of uses for cannabinoids that we need to explore.

Leave a Comment